CROSSJECT est une société pharmaceutique de spécialités qui révolutionne l’administration de médicaments d’urgence à action rapide grâce à ZENEO®, son auto-injecteur sans aiguille sûr et fiable dédié aux situations d’urgence.






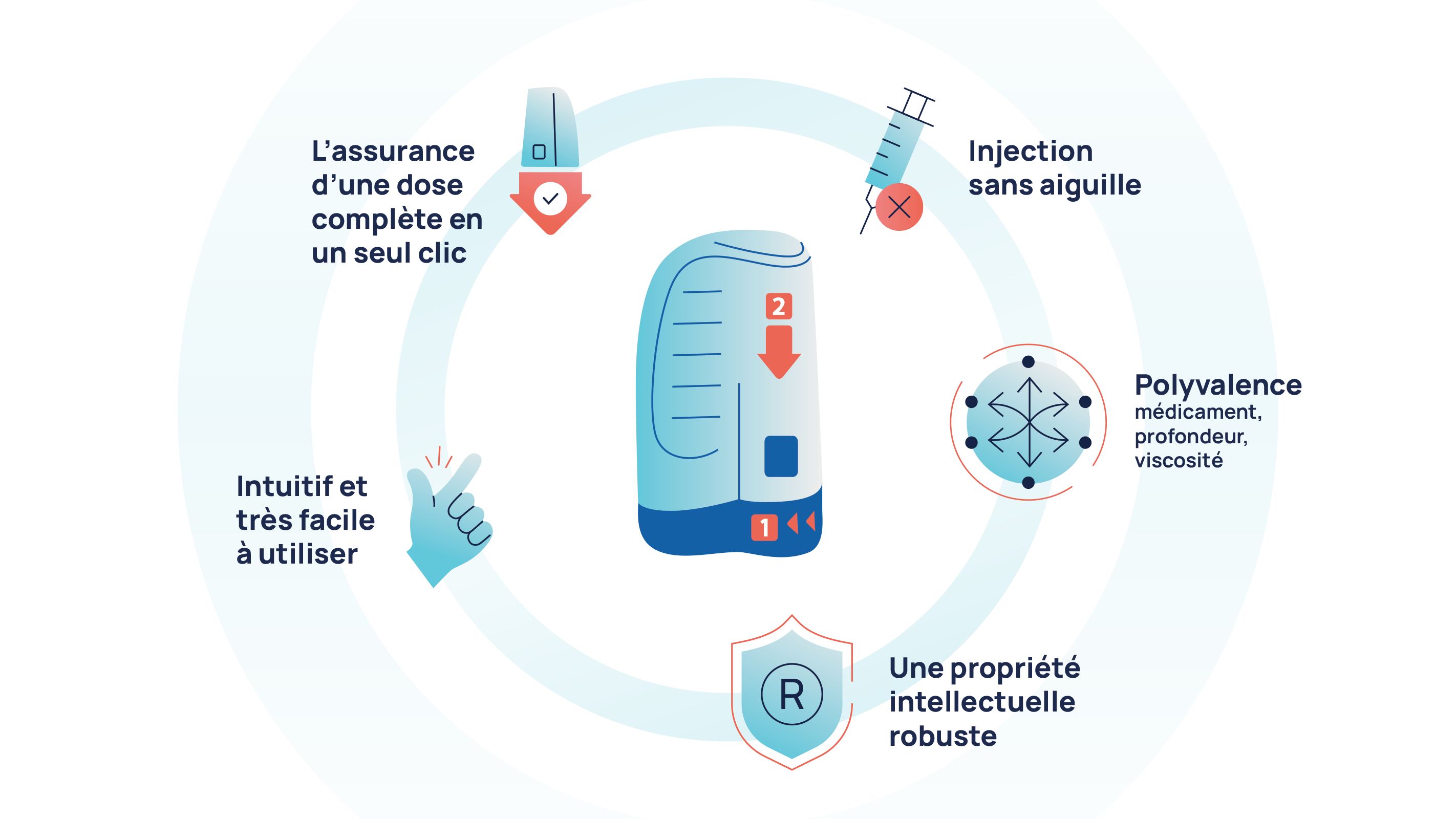
SANS AIGUILLE, RAPIDE, SÛR ET TRÈS FACILE A UTILISER
La technologie ZENEO® est le fondement de nos solutions innovantes
Aires thérapeutiques
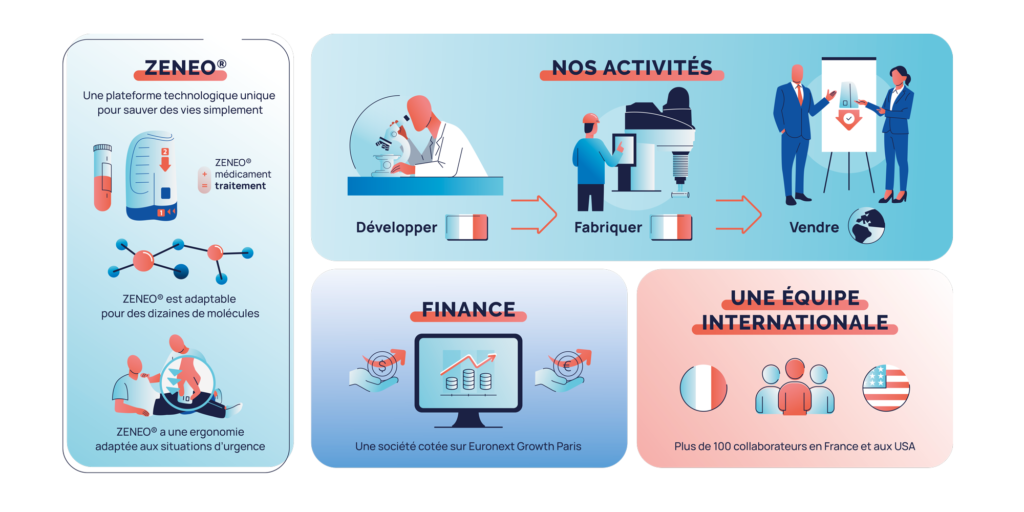
A PROPOS DE CROSSJECT




Copyright © 2025 CROSSJECT